Mi-Trial For Academia
Built for Research
Mi-Trial supports academic research with study apps to help participants stay informed, organised, and engaged throughout their study journey. The Mi-Trial app offers a single repository for all of a participant’s study information. Mi-Trial provides clear study scheduling and timely communication to reduce missed visits, support protocol adherence, and minimise administrative burden.
Why Academics Choose Mi-Trial
-
Cost‑effective for grant‑funded studies
We understand the constraints of academic budgets. Mi‑Trial offers transparent, predictable pricing which fits within typical grant structures - without compromising on quality or compliance.
-
Designed with governance in mind
Mi‑Trial supports the requirements of NHS information governance which makes it easier for investigators to secure approvals and progress quickly.
-
Proven in real-world research
Our platform underpins studies such as CF Tracker, developed and deployed in academic environments.
-
Flexible and configurable
Mi‑Trial adapts to your study.
Case Study (The CF-Tracker Study)
Mi-Trial is providing the study app for a major new study investigating the causes of lung infections in people with Cystic Fibrosis. The CF-Tracker study is sponsored by the University of Manchester and funded by the CF Trust and the medical charity LifeArc as part of the work of the Pulse-CF Innovation Hub they fund at University of Manchester.

In order to support the study, Mi-Trial has provided a standalone version of the app, rebadged and branded as the CF-Tracker app. This contains all the usual features of Mi-trial, as well some additional features. The study involves sending home samples and questionnaires every 2 weeks, so the diary function and reminders are an essential part of this. In this version, Mi-trial have added an extra feature: the app provides a unique link to a Patient Reported Outcome Measure (PROM). The PROM is hosted on SnapSurvey, so makes use of their extensive experience and well-established information governance. The app provides a link that only goes live during the assessment window, and links out seamlessly on the phone to the CF-Tracker branded PROM, with hidden fields linking to the responders study ID.
Creating this version of Mi-Trial adds another layer of functionality to Mi-Trial. It was also essential to the CF-Tracker study, which is now up and running in 18 UK CF centres. Patient feedback has been really positive and the team are delighted with their new study-specific app.
Find out more: https://www.pulse-cf.com/tracker-study
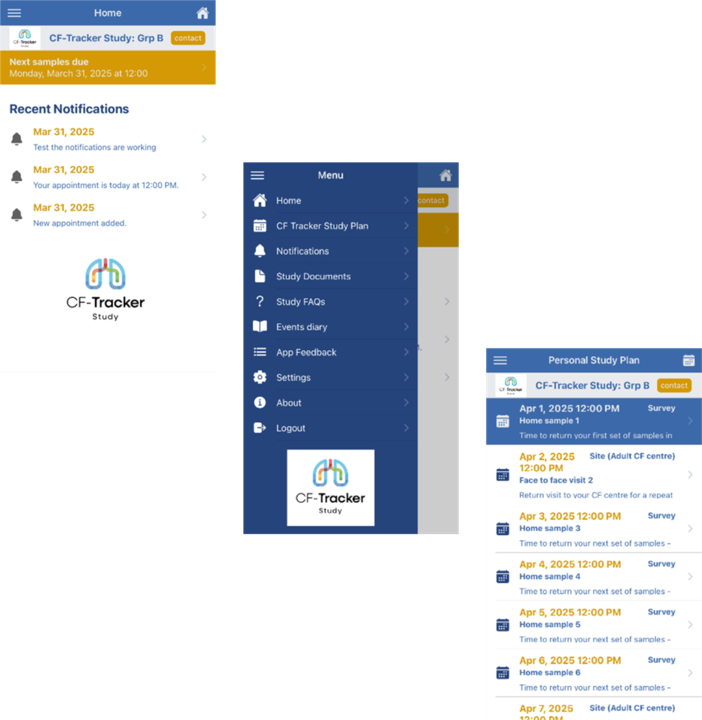
Participants have the option of using the CF-Tracker study app, downloadable from Android or Apple. The app:
- Includes all study information to access easily.
- Shows diary of reminders and notifications.
- Provides a link to the questionnaire for that week.
Assurance
Mi-Trial has been designed specifically to support use in academic research studies and has been developed in-line with established regulatory, data protection, and information governance requirements. It is intended to act as a participant-facing study coordination and communication tool, operating alongside and not replacing existing clinical, research, or data capture systems.
UK GDPR
Mi-Trial operates in accordance with data protection principles. Key features include:
- Data minimisation: only data required for study delivery and coordination is collected.
- Clear participant-facing privacy information within the app.
- Defined roles / responsibilities between study sponsor, research site, and platform provider.
- No secondary use of participant data.
Mi-Trial does not use participant data for purposes outside the delivery of the approved study. Supporting documentation (Privacy Notice, Privacy Policy, Terms & Conditions, Patient and Clinician EULAs) is available to support ethics, R&D, and information governance review.
US CFR 21 Part 11
Mi-Trial has been developed in line with the principles of CFR 21 Part 11 relating to electronic records and auditability. This includes:
- Controlled user access and permissions.
- Secure system authentication.
- Comprehensive audit logging of relevant system actions. Mi-Trial maintains audit logs for key system activities, including: study configuration changes, user actions within the researcher portal, and updates to participant study schedules and instructions.
- Documented development, testing, and validation activities.
Hosting
Mi-Trial is hosted on secure, UK-based infrastructure appropriate for health and academic research use. The platform incorporates:
- Role-based access controls.
- Secure authentication for research staff and participants (MFA).
- Separation between participant-facing and researcher-facing environments.
- Controlled deployment and change management processes.
- Access to the Mi-Trial portal is restricted to authorised study personnel only.
Cost
Mi-Trial is provided to academic research studies using a study-based costing model designed to support grant-funded research. Costs are proportionate to the scale and complexity of each study and are suitable for inclusion within standard academic funding applications.
Pricing is typically based on the duration of the study, the number of participants and sites involved, and the level of study configuration and support required. Study costs may include initial study setup and configuration, participant and research team access to the Mi-Trial platform, and ongoing technical support for the duration of the study.
Mi-Trial costs can be included within grant applications as a digital study support or infrastructure cost. A detailed, study-specific costing breakdown can be provided to support grant submissions, sponsor approvals, and local R&D or finance review.
Our modular design and proven track record means we can provide rapid, low-cost options immediately as well as more complex study-specific apps faster and cheaper than competitors.
For further information please contact hello@mi-trial.com.
Frequently Asked Questions (FAQs)
What types of studies is Mi-Trial suitable for?
Mi-Trial can be used in interventional and observational academic studies, including NIHR-supported research, single- and multi-centre studies, and longitudinal or hybrid digital studies. The platform is condition-agnostic and configurable per study.
Does Mi-Trial replace EDC platforms?
No. Mi-Trial does not collect research outcome data and is not an EDC or PRO platform. It is designed to operate alongside existing systems, focusing on study scheduling, participant instructions, reminders, and engagement.
How is Mi-Trial used by participants?
Participants access Mi-Trial via a mobile app where they can view a personalised study plan, receive reminders and instructions, access study documents, and find contact details for the research team. This helps participants understand what is expected and when.
How is Mi-Trial used by research teams?
Research teams use a secure web-based portal to configure study schedules and visits, manage participant communications, and monitor progress against the study plan. Access is role-based and restricted to authorised study staff.